chemical structure and functional group of acetone:
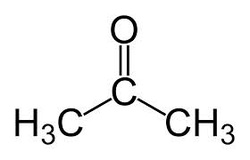 Structural Diagram of Acetone
Structural Diagram of Acetone
Acetone, an organic compound, is chemically described as CH3COCH3, and can also be referred to as 2-propanone. Acetone is a hydrocarbon derivative, more specifically a ketone in its simplest form because of its particular functional group. A ketone has a carbon double bonded to an oxygen and a bond with two other separate carbon atoms. Although similar, ketones are separate from aldehydes as aldehyde's carbonyl group is located at the end of the carbon chain, meaning that a carbon is double bonded to an oxygen, bonded to another carbon, and bonded to a hydrogen atom.
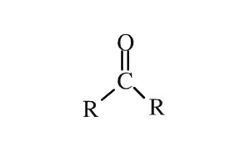 Carbonyl Group
Carbonyl Group
Acetone's functional group is the carbonyl group, which is represented by the double bond between the carbon and oxygen atom. The functional group is what gives ketone's their physical and chemical properties.
Properties of Acetone:
|
Physical:
|
Chemical:
|
Acetone's functional group, being the carbonyl group, is polar. This is because the electronegativity of the oxygen is greater than that of the carbon, creating a polar bond. The carbonyl group interacts with water, allowing it to be able to participate in hydrogen bonding. Although ketone's cannot hydrogen bond with themselves, they can with water molecules. This, as well as their polarity, gives them the property of being able to disassociate in water.
Acetone's melting and boiling points are much lower than that of waters, because acetone does not have as strong of intermolecular forces as water does. Because of the weaker forces of attraction between acetone molecules and the greater intermolecular forces between water due to its ability to hydrogen bond, less energy is needed to separate the acetone molecules and break them down.
Acetone's melting and boiling points are much lower than that of waters, because acetone does not have as strong of intermolecular forces as water does. Because of the weaker forces of attraction between acetone molecules and the greater intermolecular forces between water due to its ability to hydrogen bond, less energy is needed to separate the acetone molecules and break them down.
Chemical reactions involving acetone:
Acetone is a versatile compound, having the ability to undergo addition, oxidation, reduction, and condensation reactions. It is also used as a raw material in chemical synthesis, roughly 75% of the available acetone is used to produce other chemicals.
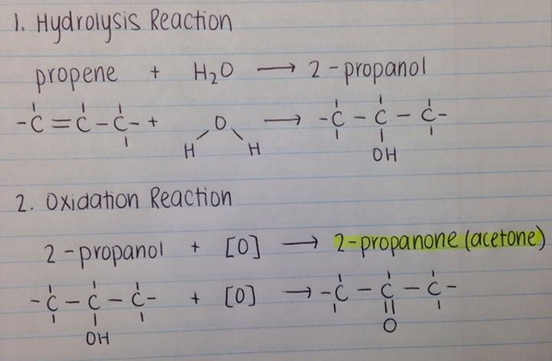
Synthesis of Acetone from an Alkene:
Synthesis of Acetone from an Alkene:
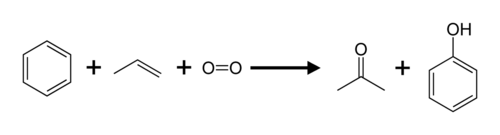
Acetone Produced from the Cumene Process:
Approximately 83% of acetone is produced from the cumene process. The cumene process is an industrial process for developing phenol and acetone from benzene and propene. During the cumene process, benzene is alkylated with propene to produce cumene, which is then oxidized by air to produce phenol and acetone.
Approximately 83% of acetone is produced from the cumene process. The cumene process is an industrial process for developing phenol and acetone from benzene and propene. During the cumene process, benzene is alkylated with propene to produce cumene, which is then oxidized by air to produce phenol and acetone.